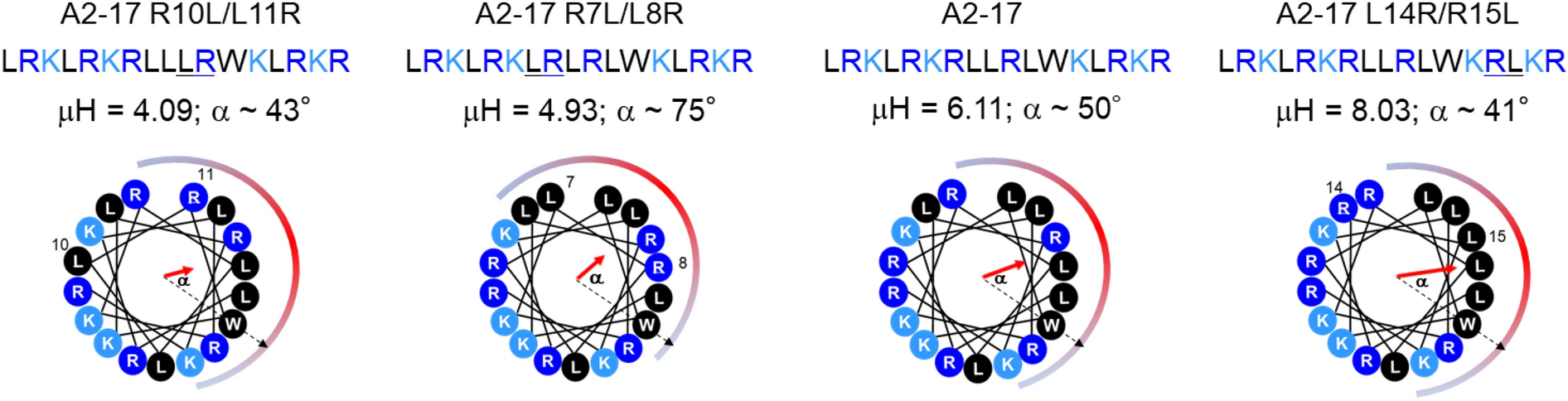
Analysis of hydrophobic and hydrophilic moments of short penetrating peptides for enhancing mitochondrial localization: prediction and validation - Pirisinu - 2019 - The FASEB Journal - Wiley Online Library
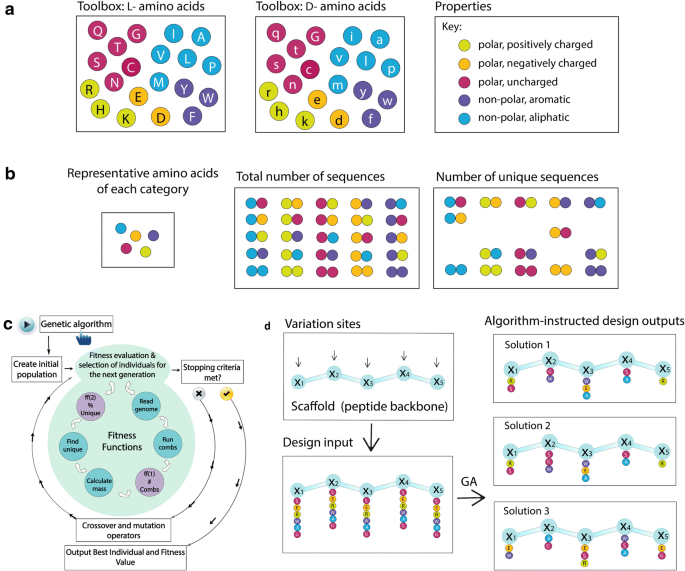
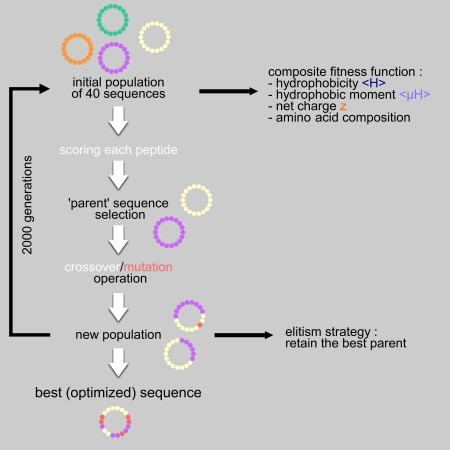
Algorithm-supported, mass and sequence diversity-oriented random peptide library design | Journal of Cheminformatics | Full Text

Mean hydrophobicity, hydrophobic moment, net charge and helical wheel... | Download Scientific Diagram
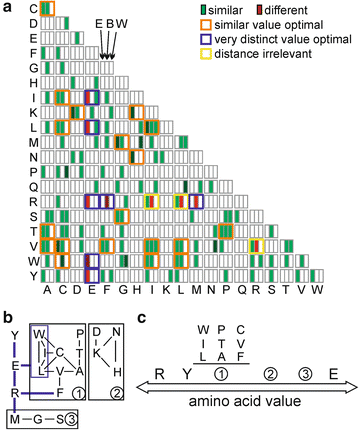
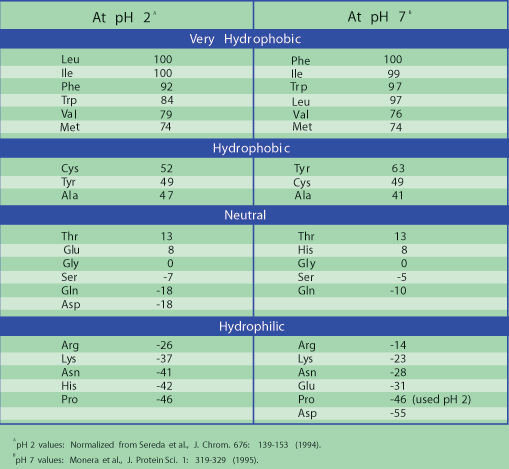
50 years of amino acid hydrophobicity scales: revisiting the capacity for peptide classification | Biological Research | Full Text

Efficient transdermal delivery of functional protein cargoes by a hydrophobic peptide MTD 1067 | Scientific Reports

Connecting Hydrophobic Surfaces in Cyclic Peptides Increases Membrane Permeability - Hoang - 2021 - Angewandte Chemie International Edition - Wiley Online Library

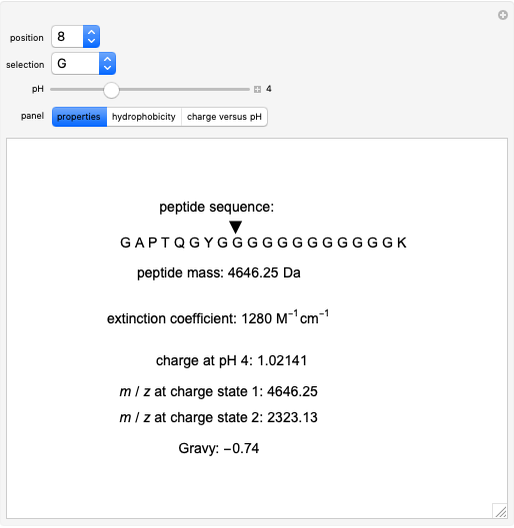
Bachem Group - Calculate your Peptide. Our Peptide Calculator is an online tool that calculates molecular weight, net charge, isoelectric point, and average hydrophobicity for your peptide sequence. Try it out. http://www.bachem.com/service-support ...

Characterizing hydrophobicity of amino acid side chains in a protein environment via measuring contact angle of a water nanodroplet on planar peptide network | PNAS
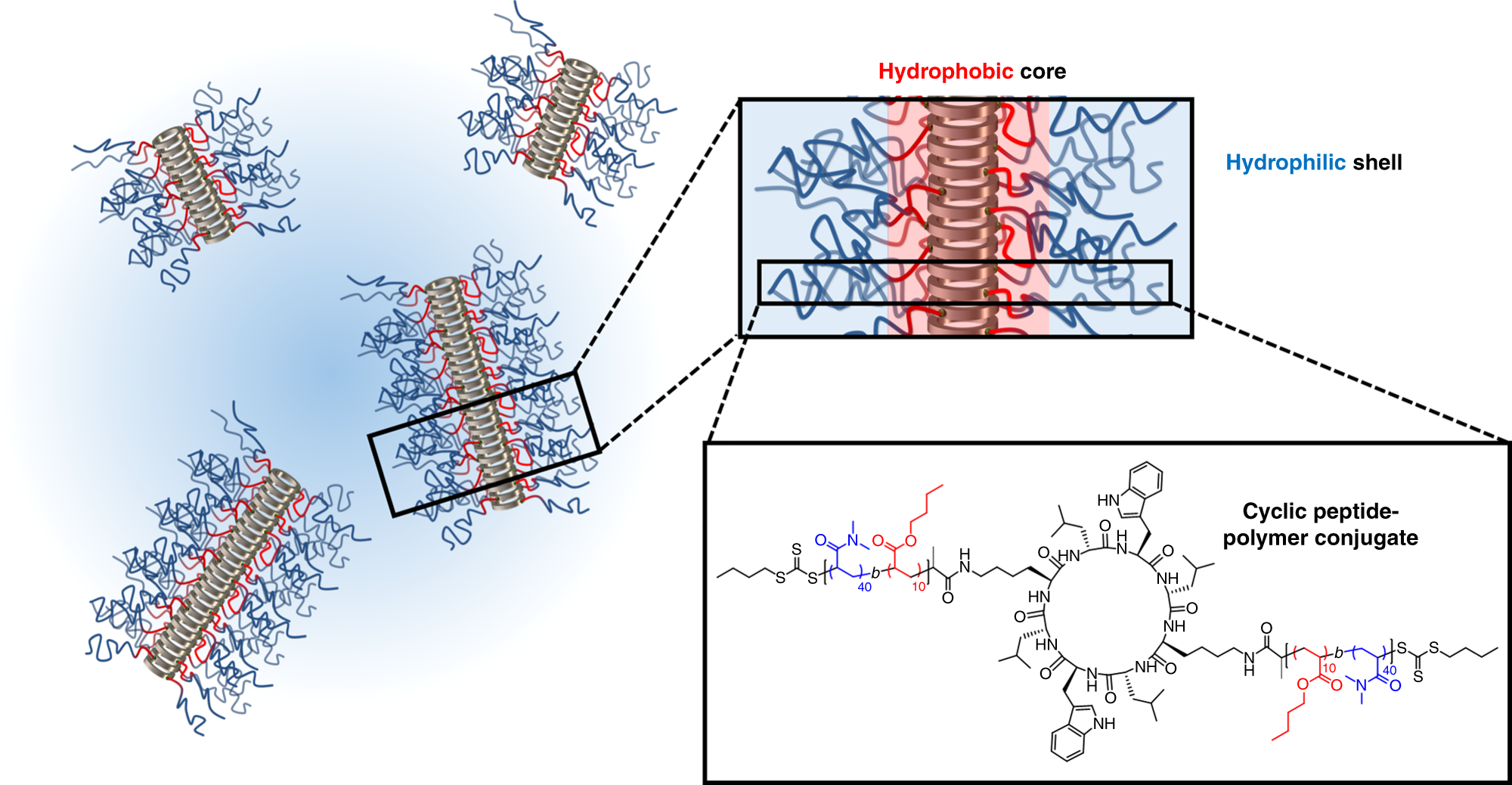
Dual self-assembly of supramolecular peptide nanotubes to provide stabilisation in water | Nature Communications

Characterizing hydrophobicity of amino acid side chains in a protein environment via measuring contact angle of a water nanodroplet on planar peptide network | PNAS

Analysis of hydrophobic and hydrophilic moments of short penetrating peptides for enhancing mitochondrial localization: prediction and validation - Pirisinu - 2019 - The FASEB Journal - Wiley Online Library

Equation 1 was used to determine effective hydrophobicity. The helical... | Download Scientific Diagram

Interactions of Cationic-Hydrophobic Peptides with Lipid Bilayers: A Monte Carlo Simulation Method: Biophysical Journal

What can machine learning do for antimicrobial peptides, and what can antimicrobial peptides do for machine learning? | Interface Focus

Methods of calculating protein hydrophobicity and their application in developing correlations to predict hydrophobic interaction chromatography retention - ScienceDirect