
an empirical formula C3H3O if molar mass of the compound 110.15 molecular formula of organic compound is? - Brainly.in

Calculate the mass of compound (molar mass = 256 g mol^-1 ) to be dissolved in 75g of benzene to lower its freezing point by 0.48 K (Kf = 5.12 K kg mol^-1 ) .

Calculate the molar mass of the following substances. (a) Ethyne, C2H2 (b) Sulphur molecule, S8 (c) Phosphorus molecule, P4 (Atomic mass of phosporus = 31 ) (d) Hydrochloric acid, HCl (e) Nitric acid, HNO3



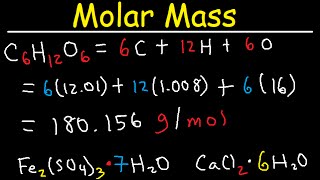




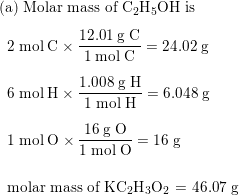









![Example] How to Find the Molar Mass of a Compound. - YouTube Example] How to Find the Molar Mass of a Compound. - YouTube](https://i.ytimg.com/vi/_qPeh8bwkiw/maxresdefault.jpg)


