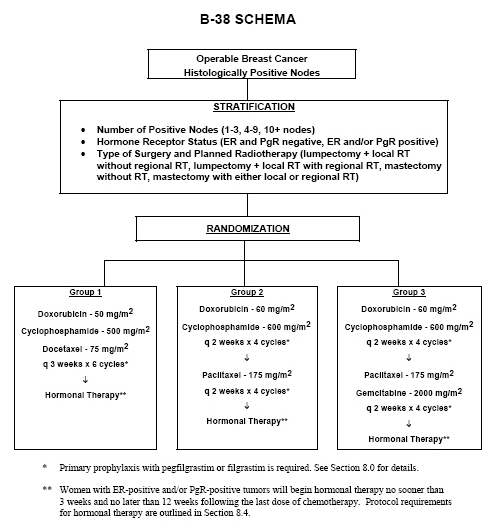
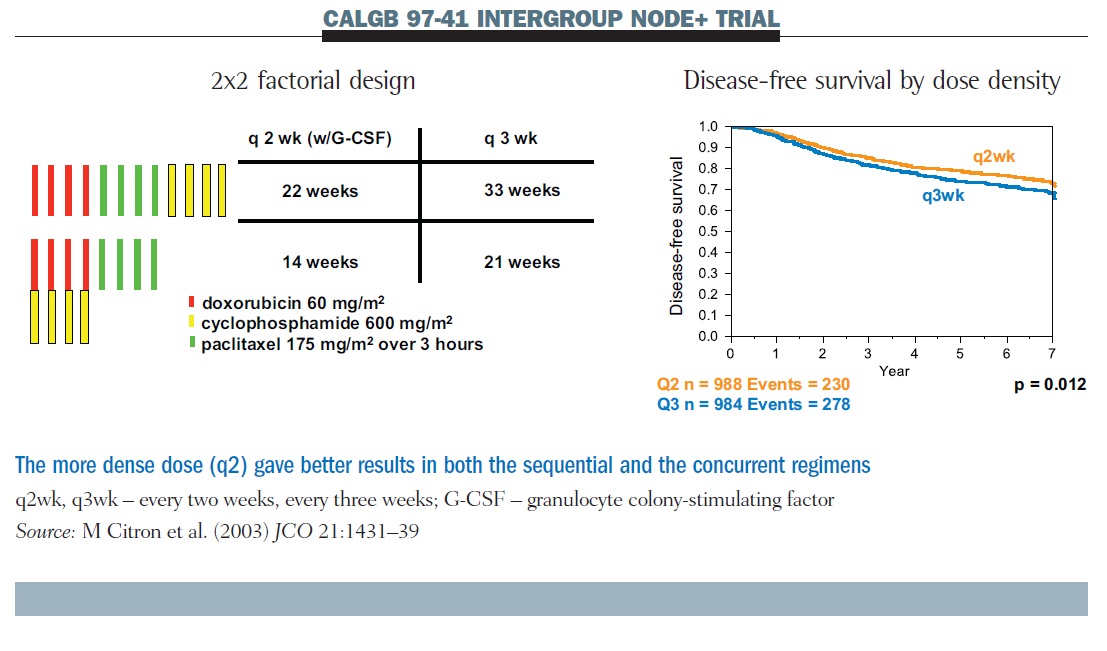
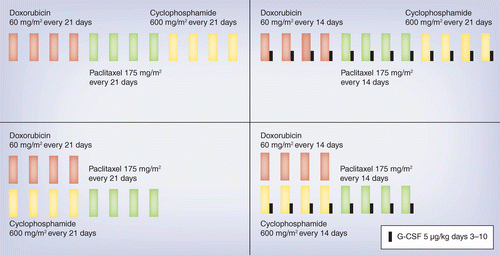
Four cycles of docetaxel and cyclophosphamide as adjuvant chemotherapy in node negative breast cancer: A real-world study - ScienceDirect

Pertuzumab, trastuzumab, and standard anthracycline- and taxane-based chemotherapy for the neoadjuvant treatment of patients with HER2-positive localized breast cancer (BERENICE): a phase II, open-label, multicenter, multinational cardiac safety study ...

Anthracycline-free or short-term regimen as adjuvant chemotherapy for operable breast cancer: A phase III randomized non-inferiority trial - The Lancet Regional Health – Western Pacific

Phase 2 Study of Dose-Dense Doxorubicin and Cyclophosphamide Followed by Eribulin Mesylate With or Without Prophylactic Growth Factor for Adjuvant Treatment of Early-Stage Human Epidermal Growth Factor Receptor 2–Negative Breast Cancer -
BC Cancer Protocol Summary for NEOAdjuvant Therapy for Breast Cancer using Dose Dense Therapy: PACLitaxel followed by DOXOrubic

Standardizing Chemotherapy Regimen Nomenclature: A Proposal and Evaluation of the HemOnc and National Cancer Institute Thesaurus Regimen Content | JCO Clinical Cancer Informatics
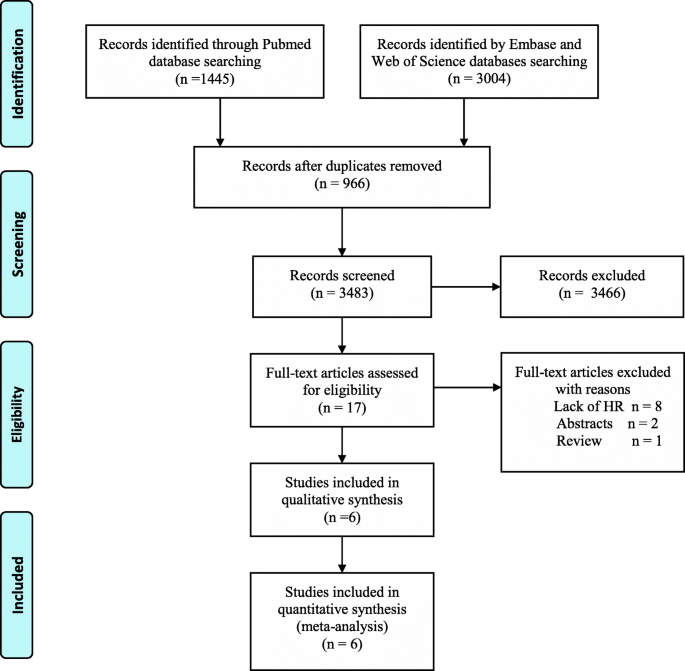
Efficacy of chemotherapy for lymph node-positive luminal A subtype breast cancer patients: an updated meta-analysis | World Journal of Surgical Oncology | Full Text
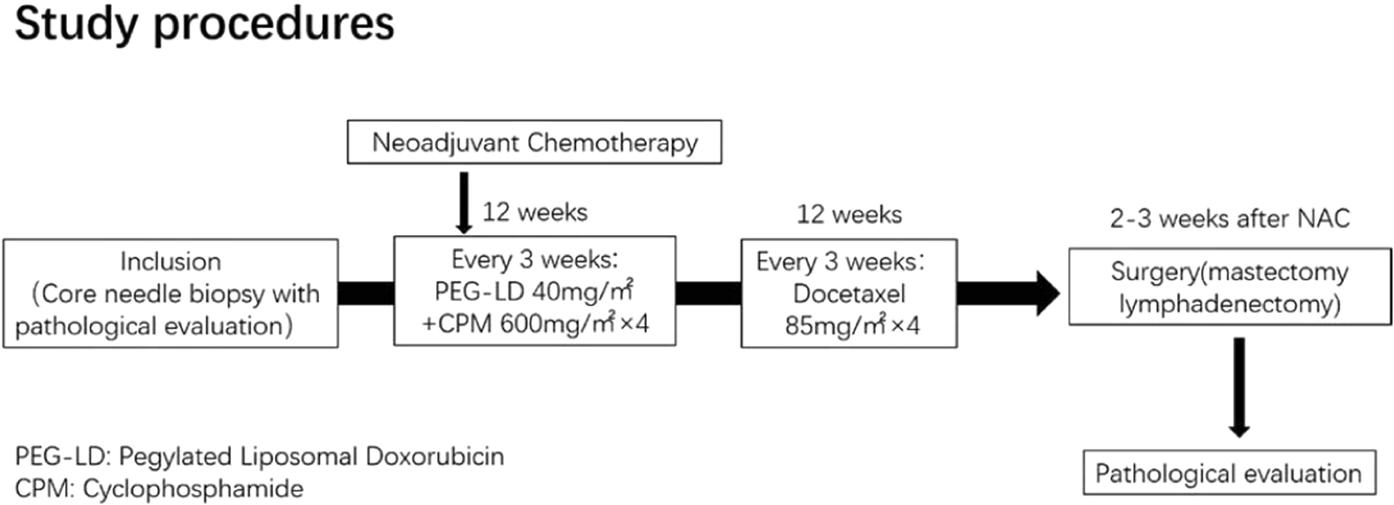
Pegylated liposomal doxorubicin plus cyclophosphamide followed by docetaxel as neoadjuvant chemotherapy in locally advanced breast cancer (registration number: ChiCTR1900023052) | Scientific Reports
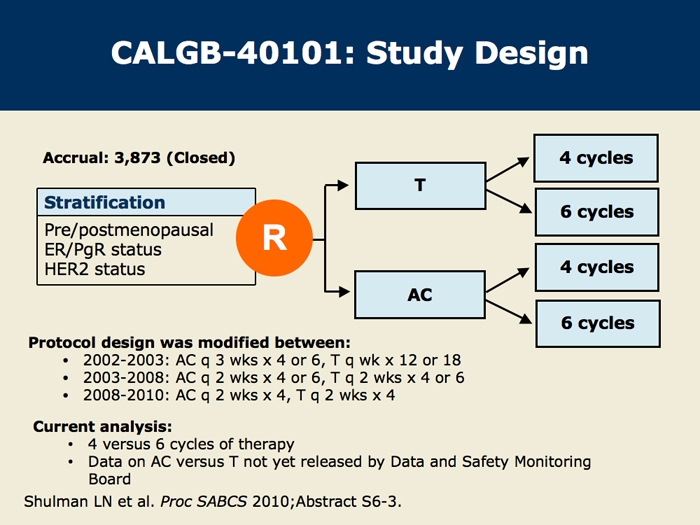
Effect of Number of Treatment Cycles of Adjuvant Chemotherapy on Clinical Outcomes | Research To Practice